-
Things to Do
Discover everything to do in University City.
-
 Community CalendarEvents including restaurants promotions, concerts, live theater, community meetings, art galleries & public spaces
Community CalendarEvents including restaurants promotions, concerts, live theater, community meetings, art galleries & public spaces -
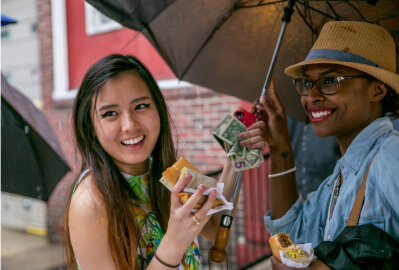 Baltimore Avenue Dollar Stroll$1, $3, and $5 deals from local businesses & vendors
Baltimore Avenue Dollar Stroll$1, $3, and $5 deals from local businesses & vendors -
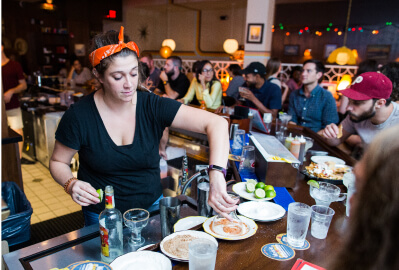 University City Dining DaysUniversity City’s version of “restaurant week”
University City Dining DaysUniversity City’s version of “restaurant week” -
 University City Summer SeriesFamily-friendly free outdoor summer concerts
University City Summer SeriesFamily-friendly free outdoor summer concerts -
 Movies in Clark ParkFamily-friendly free outdoor screenings every August
Movies in Clark ParkFamily-friendly free outdoor screenings every August -
 University City MixNetworking happy hour series
University City MixNetworking happy hour series
- About the Neighborhood
Learn More About our Thriving Neighborhood.-
Neighborhood OverviewUniversity City is Philadelphia’s academic heart, a hub for innovation & scientific discovery, and a historic neighborhood full of artistic & cultural exploration.
-
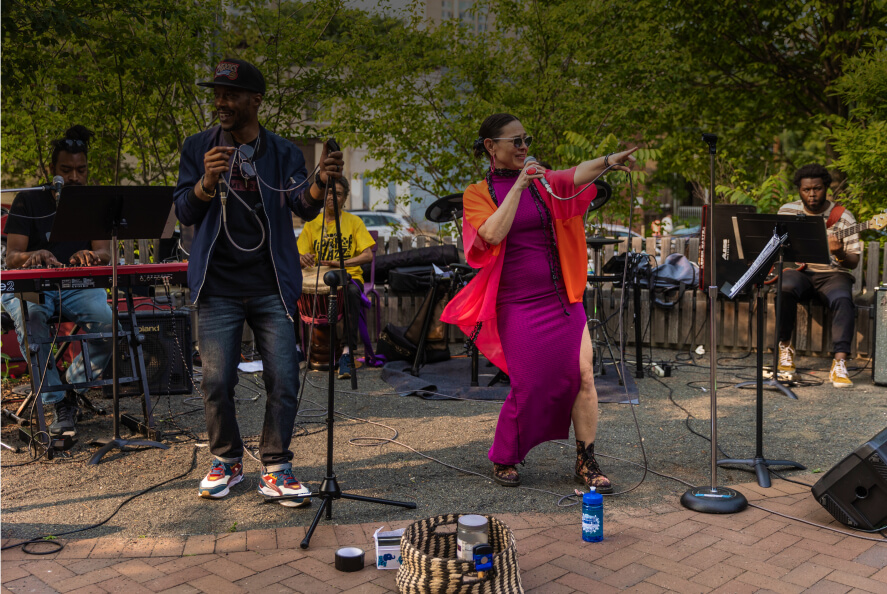 Neighborhood SpotlightsExplore top food, art, shopping, and culture, just minutes from Center City.
Neighborhood SpotlightsExplore top food, art, shopping, and culture, just minutes from Center City. -
 Latest NewsUpdated on UCD's work and the neighborhood.
Latest NewsUpdated on UCD's work and the neighborhood.
- Doing Business
Learn How we Promote Growth and Innovation.-
 Choose University CityUniversity City is a top Philadelphia neighborhood to work, live, attend school, open a business, or visit.
Choose University CityUniversity City is a top Philadelphia neighborhood to work, live, attend school, open a business, or visit. -
Reports and DataResearch & analysis tied to the local economy, transportation, public space use, public safety, workforce development, & more.
-
 Become a SponsorSponsorships provide valuable visibility while fostering community engagement, and enhancing the vibrancy of the neighborhood.
Become a SponsorSponsorships provide valuable visibility while fostering community engagement, and enhancing the vibrancy of the neighborhood.
- What We Do
Explore our Range of Services and Specialties.-
 Maintain Clean and Safe StreetsCreating a safe and welcoming environment for residents, employees, students, and visitors
Maintain Clean and Safe StreetsCreating a safe and welcoming environment for residents, employees, students, and visitors -
 Assist the UnhousedAddressing the tragedy of homelessness in our district through a compassionate, measured & people-first approach
Assist the UnhousedAddressing the tragedy of homelessness in our district through a compassionate, measured & people-first approach -
 Improve TransportationAddressing bike & pedestrian safety, traffic congestion & public transit routing
Improve TransportationAddressing bike & pedestrian safety, traffic congestion & public transit routing -
 Support Our Local EconomySupporting supports the local business community and residents through a wide range of services
Support Our Local EconomySupporting supports the local business community and residents through a wide range of services -
 Provide Landscape ServicesOur landscaping social venture provides design-build-maintenance landscaping services
Provide Landscape ServicesOur landscaping social venture provides design-build-maintenance landscaping services -
 Connect Residents to CareersHarnessing the hiring power of our area’s major employers while cultivating local talent
Connect Residents to CareersHarnessing the hiring power of our area’s major employers while cultivating local talent
-


-
Things to Do
Discover everything to do in University City.
-
Community CalendarEvents including restaurants promotions, concerts, live theater, community meetings, art galleries & public spaces
-
Baltimore Avenue Dollar Stroll$1, $3, and $5 deals from local businesses & vendors
-
University City Dining DaysUniversity City’s version of “restaurant week”
-
University City Summer SeriesFamily-friendly free outdoor summer concerts
-
Movies in Clark ParkFamily-friendly free outdoor screenings every August
-
University City MixNetworking happy hour series
- About the Neighborhood
Learn More About our Thriving Neighborhood.-
Neighborhood OverviewUniversity City is Philadelphia’s academic heart, a hub for innovation & scientific discovery, and a historic neighborhood full of artistic & cultural exploration.
-
Neighborhood SpotlightsExplore top food, art, shopping, and culture, just minutes from Center City.
-
Latest NewsUpdated on UCD's work and the neighborhood.
- Doing Business
Learn How we Promote Growth and Innovation.-
Choose University CityUniversity City is a top Philadelphia neighborhood to work, live, attend school, open a business, or visit.
-
Reports and DataResearch & analysis tied to the local economy, transportation, public space use, public safety, workforce development, & more.
-
Become a SponsorSponsorships provide valuable visibility while fostering community engagement, and enhancing the vibrancy of the neighborhood.
- What We Do
Explore our Range of Services and Specialties.-
Maintain Clean and Safe StreetsCreating a safe and welcoming environment for residents, employees, students, and visitors
-
Assist the UnhousedAddressing the tragedy of homelessness in our district through a compassionate, measured & people-first approach
-
Improve TransportationAddressing bike & pedestrian safety, traffic congestion & public transit routing
-
Support Our Local EconomySupporting supports the local business community and residents through a wide range of services
-
Provide Landscape ServicesOur landscaping social venture provides design-build-maintenance landscaping services
-
Connect Residents to CareersHarnessing the hiring power of our area’s major employers while cultivating local talent

Latest News

-
